China’s supplement market is booming—and the regulatory environment is changing fast. Under GACC Order 280, effective June 1, 2026, the rules for overseas supplement manufacturers are shifting. Whether you sell via cross-border e-commerce (CBEC), general trade, or C2C parcels, understanding the compliance landscape is essential to avoid costly rejections.
This guide provides a complete roadmap for importing supplements into China across all three channels, tailored specifically for health product brands.
Why China? The Supplement Market Opportunity
China’s dietary supplement market has grown steadily over the past decade. In 2025, the market reached approximately 312.4 billion RMB, growing at a compound annual rate of 8.65% since 2016. The broader nutritional health product market reached 447.7 billion RMB in 2025, with an average annual growth of 8.3%. Within this market, vitamin and mineral products dominate—accounting for 55.4% of sales in 2025—while Chinese herbal supplements have surged to 99.2 billion RMB, driven by the “food and medicine homology” culture.
Drivers include rapid population aging, rising health awareness among younger consumers, ongoing policy support, and the continued expansion of cross-border e-commerce channels.
Three Paths to Market: At a Glance
| Feature | CBEC | General Trade | C2C |
| Blue Hat required | ❌ No | ✅ Yes | ❌ No |
| GACC registration required | ❌ No | ✅ Yes | ❌ No |
| Chinese labeling | ❌ Not on product | ✅ Required | ❌ Not required |
| Tax rate (typical supplement) | ~9.1% | 20–35% | 13% |
| Entry time | Weeks | 2–5 years | Days |
| Sales channels | CBEC platforms only | All channels | Personal use only |
The table above highlights the most striking difference: general trade requires both Blue Hat certification and GACC registration, while CBEC offers a streamlined path with neither. Let’s examine each channel in depth.
🇨🇳 Part 1: General Trade – The Traditional Path
General trade treats supplements as regulated health products entering China for general consumption. This path requires two separate certifications.
Blue Hat (蓝帽子) Registration
“Blue Hat” is the mandatory approval for health foods sold through domestic channels in China—administered by the State Administration for Market Regulation (SAMR).
Two pathways under Blue Hat:
| Pathway | Products | Timeline | Cost |
| Registration | Products with specific health claims (immune support, memory enhancement) | 2–5 years | $50,000–200,000+ per product |
| Filing | Vitamins, minerals, and other nutrients listed in official catalogs | 6–12 months | Lower |
Once approved, registration certificates are valid for five years and may be renewed.
GACC Registration Under Order 280
Under GACC Order 280 (effective June 1, 2026), overseas supplement manufacturers exporting to China must register with GACC’s CIFER system. Supplement products require “official recommendation” registration, meaning your home country’s competent authority must first approve your facility and issue a recommendation letter.
Key GACC requirements for supplements:
- Submit enterprise registration application and identity proof
- Submit compliance statement with Chinese food safety standards
- For official recommendation products, provide home country review report and recommendation letter
- Receive GACC registration number for packaging
⚠️ Critical distinction: General trade supplements require both Blue Hat (from SAMR) and GACC manufacturer registration. Having a Blue Hat alone does not allow import—the manufacturer must also have a GACC registration number.
Labelling Requirements
General trade supplements must have Chinese-language labels on each unit, including:
| Required Information | Details |
|---|---|
| Blue Hat logo | Required on all domestic health foods |
| Product name (approved Chinese name) | Must match registration certificate |
| Ingredients and net content | Full disclosure per GB standards |
| Health functions | Limited to officially approved claims |
| Shelf life and storage conditions | With production date |
| Importer information | Name and address |
Tax Calculation (General Trade)
General trade taxes combine customs duty (MFN rate, typically 5–25% depending on the product), VAT (food products: 9% or 13%), and consumption tax where applicable. Total effective tax rates often range from 20% to 35% or higher, compared to ~9% for CBEC.
🛒 Part 2: CBEC – The Fast Track for Supplements
For overseas supplement brands, CBEC is typically the preferred entry path. Health foods and supplements can be exported via CBEC without Blue Hat registration. However, GACC registration may still be required.
Why CBEC Works for Supplements
China offers two fundamentally different regulatory frameworks for health supplements: domestic (requiring Blue Hat) and cross-border (CBEC). CBEC is the path most foreign brands choose because it provides faster market access, lower compliance burden, and lower taxes.
GACC Order 280 and CBEC Supplements
Does GACC Order 280 affect CBEC supplements? The short answer is No. Multiple sources confirm that the regulation does not directly impact cross-border dietary supplements, and merchants can temporarily rest assured that CBEC remains outside the general trade registration framework.
However, Article 30 of Order 280 states that CBEC requirements “shall be handled in accordance with relevant provisions”—a potential signal that the exemption could be reviewed in the future. Hence, company should always be prepared to obtain GACC certification.
CBEC Tax Rates for Supplements
Under CBEC, products benefit from preferential tax treatment:
- Customs duty: 0%
- VAT: 70% of standard rate (approx. 9.1% for most supplements)
- Consumption tax: 70% of applicable rate
The CBEC comprehensive tax for most supplements is approximately 9.1% —significantly lower than general trade.
📌 Real-World Calculation: For a $100 supplement import via CBEC: duty $0, VAT ~$9.10, total ~$9.10.
Order Limits
| Limit Type | Amount |
|---|---|
| Per transaction | ≤ 5,000 RMB |
| Per consumer annually | ≤ 26,000 RMB |
Orders exceeding 5,000 RMB but within the annual limit are subject to general trade duties on the excess portion.
Labelling Requirements
CBEC supplements do not require Chinese labels on the physical product. However, the ordering webpage must provide Chinese translation of product information, including ingredients, usage instructions, and allergen warnings.
CBEC Positive List
To qualify for CBEC import, your supplement’s HS code must appear on the CBEC Positive List (跨境电子商务零售进口商品清单). The 2025 version includes 1,476 8-digit HS codes.
Common supplement categories on the list include vitamins, minerals, amino acids, fish oil, probiotics, and herbal extracts. However, certain products remain prohibited—including meat products, sterilized milk, egg yolk, and bird’s nest products.
📌 Always verify your product’s eligibility before planning a CBEC launch.
Supplement Ingredients Under Scrutiny
Even with CBEC’s lighter regulatory touch, Chinese customs is tightening oversight on certain supplement ingredients. Recent enforcement has seen products containing veratrum (藜芦), gamma-hydroxybutyric acid, and sucralose in excessive amounts rejected.
The government has expanded its blacklist of illegal additives to include yohimbine, carboxy, sibutramine derivatives, and continues to crack down on PDE-5 inhibitors, phenolphthalein, and related substances.
Major CBEC Platforms for Supplements
| Platform | Key Features |
|---|---|
| Tmall Global | Largest CBEC platform; supplements consistently among top-selling categories |
| JD Worldwide | Strong logistics network; popular for premium and imported brands |
| Douyin Global | Livestream e-commerce; fastest-growing for discovery-driven supplement sales |
✈️ Part 3: C2C (Personal Parcels)
C2C imports—personal parcels shipped directly to individual consumers via postal or courier services—are governed by different rules.
Regulatory Framework
C2C parcels are regulated as personal-use items, not commercial cargo. No GACC registration or Blue Hat is required.
Tax Rate
C2C supplements are subject to Postal Parcel Tax (行邮税) —a combined tax covering customs duty, VAT, and consumption tax. The tax rate for food items, including supplements, is 13% .
A 50 RMB exemption applies per parcel—orders below this threshold are tax-free. However, shipments deemed commercial (exceeding “reasonable personal use” quantity) may be reclassified and face higher taxes or rejection.
Limitations
| Aspect | Details |
|---|---|
| Quantity restrictions | Strict “reasonable personal use” limits enforced |
| Business use | Not permitted; for personal consumption only |
| Best use case | Sampling, low-volume direct sales, gifts |
📊 3 Case Studies: How Supplement Brands Succeed with CBEC
Case Study 1: Swisse – Mega Brand Supplements
Swisse is an Australian vitamin and supplement powerhouse that successfully positioned itself as a “Pharma-Grade” wellness expert in China. Based on the Australian TGA certification and EU GMP compliance in 95%+ content, they successfully entered China via CBEC. Their insight: 70% of Chinese consumers discover health supplements on RedNote (Xiaohongshu) before purchasing on Douyin and marketplaces (like Tmall or JD).
Key takeaways from Swisse’s approach:
- Social-first discovery: Leveraged RedNote for product education and user-generated content
- Clear science-based positioning: Emphasized evidence-based formulation with authority endorsement for their pharma-grade products.
- Platform integration: Drove social discovery traffic in RedNote, drive sales in Douyin Store and built a private community & CRM system for personalized care, boosting member loyalty and lifetime value.
- Results: #1 in China health supplements across Tmall, JD, Douyin and VIP.com

For supplement brands, Swisse’s example shows that success depends on understanding how Chinese consumers discover products—often through social platforms first, then purchase through e-commerce.
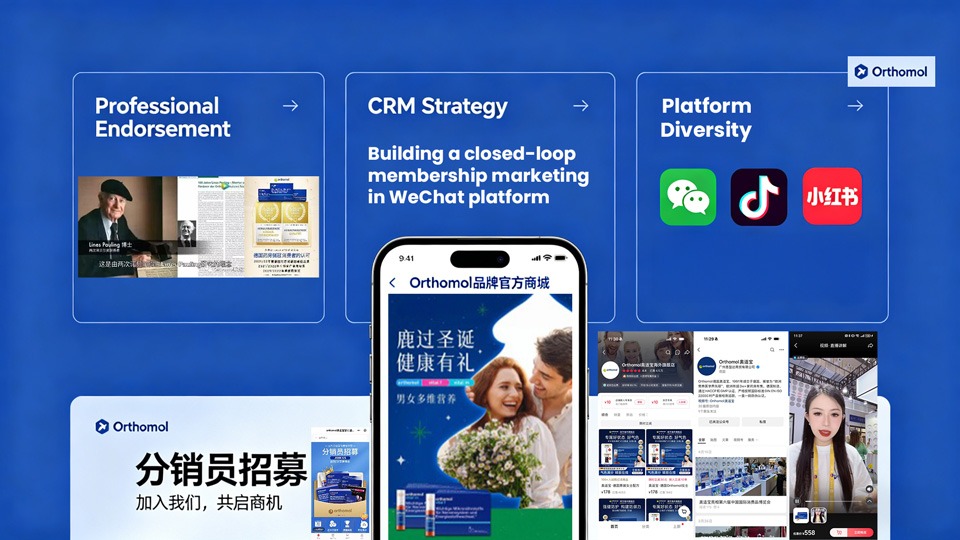
Case Study 2: Orthomol – German Luxury Supplements
Orthomol is the first supplements brand to adopt the “Orthomolecular Medicine” concept (based on research by two-time Nobel laureate Linus Pauling). This German luxury supplements demonstrated the value of premium science brand positioning and established strong growth both at home and in China.
Key takeaways from Orthomol:
- Professional Endorsement: Target nutrition, supplements and health experts to endorse their products instead of mass marketing
- CRM Strategy: Invest in building a closed-loop membership marketing in WeChat platform. Partnership with hospitals, clinics and high-end wellness centres
- Platform diversity: Apart from WeChat, the brand leverage Douyin & RedNote to collaborate with health experts often having medical or science backgrounds.
- Results: Exceptionally high customer loyalty; 200% increase in sales reported in 2023.
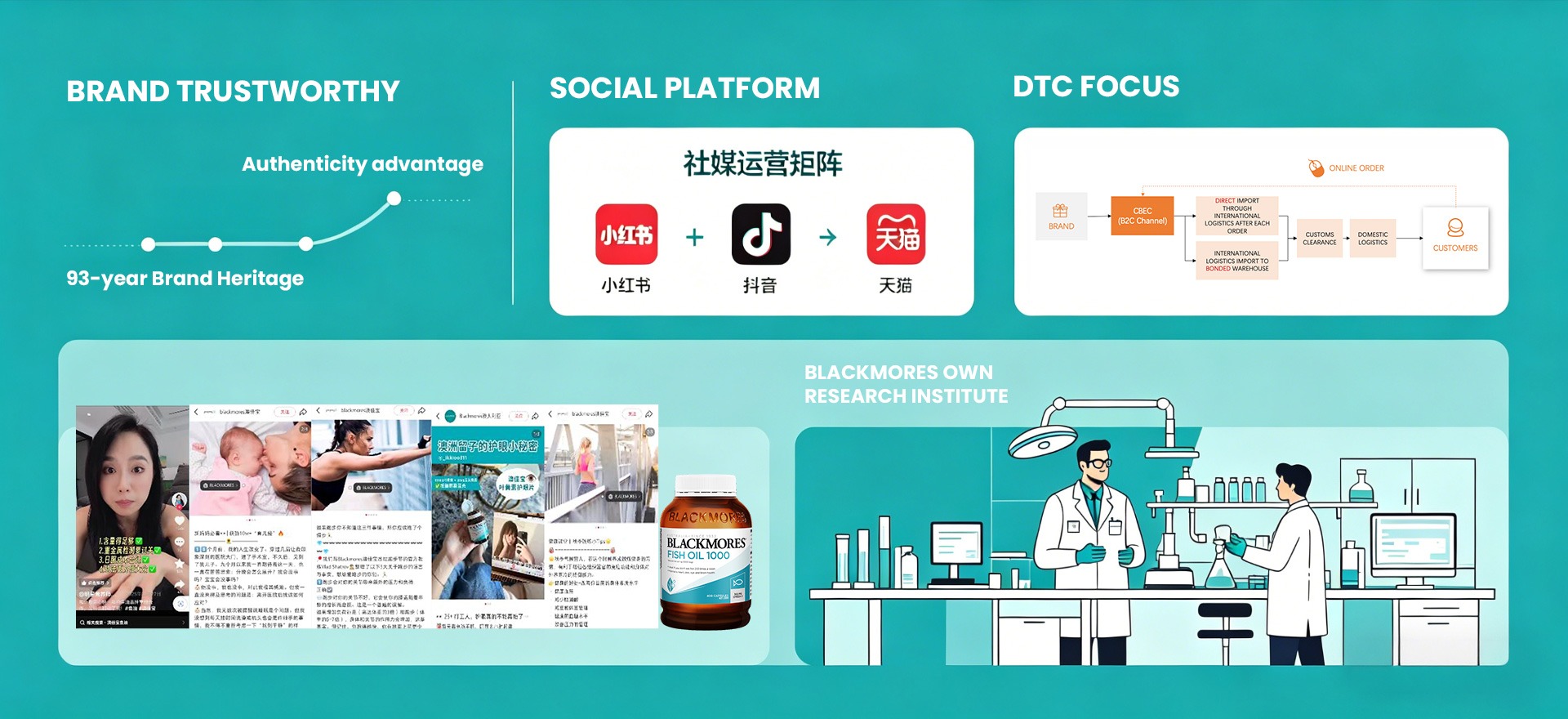
Case Study 3: Blackmores – From Pharmacy to CBEC Leader
Operates a cross-border ecommerce model, with no offline retail footprint in China, selling entirely through online platforms like Tmall Global and Douyin. Successfully builds trust on “Australian origin,” rigorous quality control (30+ tests per product), and its own research institute.
Key takeaways from Blackmores:
- Heritage & authenticity advantage: Leveraged 93-year old Australian natural health company background for credibility
- Understanding Chinese Social Media: Built brand and product content in RedNote, drive sales across RedNote, Douyin and Tmall to dominate the online market.
- Direct-to-consumer focus: Used CBEC to sell directly without intermediaries
⚖️ How to Choose Your Import Channel
| Your Situation | Recommended Channel |
|---|---|
| Testing the Chinese market, limited budget, no Blue Hat | CBEC |
| Established brand with proven demand, ready for full market access | General Trade |
| Sending samples or very low volumes to individuals | C2C (non-commercial) |
| Products with specific health claims requiring Blue Hat for general trade | General Trade |
| Products not on the Positive List | General Trade only |
| Want to sell on Tmall Global or JD Worldwide | CBEC |
| Want to sell on Douyin, RedNote, WeChat Ecommerce | CBEC |
A phased approach often works best: start with CBEC to validate demand, then pursue Blue Hat and GACC registration for general trade expansion once market success is proven. Some regions exporting to China via CBEC may require GACC registration as well.
🚫 Common Compliance Pitfalls (And How to Avoid Them)
❌ Pitfall 1: Products Not on the Positive List
The problem: HS code falls outside the Positive List → cargo returned.
How to avoid: Verify your HS code against the latest Positive List before shipping.
❌ Pitfall 2: Unapproved or Undeclared Ingredients
The problem: In February 2025 alone, 357 batches of imported food were rejected by Chinese customs. Common reasons include microbiological contamination, heavy metals, undeclared GMOs, and additive violations. Veratrum-containing products were directly rejected because this ingredient is on the prohibited list for health foods.
How to avoid: Screen all ingredients against China’s GB standards before formulation.
❌ Pitfall 3: Assuming GACC Registration Is Automatic
The problem: For general trade, missing renewal windows stops imports.
How to avoid: Track expiry dates; set reminders 12 months ahead for renewal-required categories.
❌ Pitfall 4: Misclassifying Products
The problem: Product declared as “food” when it should be “health food” → compliance gaps.
How to avoid: Understand how China classifies your product before selecting an import channel.
❌ Pitfall 5: Marketing Claims That Trigger Scrutiny
The problem: Exaggerated or unsubstantiated health claims on CBEC listings.
How to avoid: Use only compliant language; avoid claims like “cures disease.”
❌ Pitfall 6: Paying for “GACC Registration Services”
The problem: Fraudulent websites charge fees for registration.
How to avoid: GACC registration is completely free through the official portal at https://cifer.singlewindow.cn.
🔮 Ongoing Regulatory Monitoring
GACC Order 280’s CBEC exemption is currently in effect. However, the regulation states that CBEC imports “shall be handled in accordance with relevant provisions”—leaving room for future policy adjustments. Any changes to Shang Cai Fa [2018] No. 486 or GACC Announcement No. 194 of 2018 could affect the CBEC exemption.
MyMyPanda will continue to monitor official channels, including the GACC website (http://jckspj.customs.gov.cn/) and the CBEC Positive List, for any future announcements that might clarify or alter CBEC registration requirements for supplement products.
💼 How MyMyPanda Can Help
As a one-stop CBEC solution provider, MyMyPanda helps overseas supplement brands navigate every step of the China market entry process:
- ✅ CBEC Positive List verification – Ensure your products are eligible
- ✅ Platform onboarding – Tmall Global, JD Worldwide, Douyin Global
- ✅ Customs clearance and bonded warehouse logistics
- ✅ Compliance documentation preparation
- ✅ General Trade GACC registration assistance – For channel expansion
- ✅ Ongoing regulatory monitoring and updates
👉 Contact MyMyPanda Today to Start Your China Supplement Journey
Key Takeaways
| Aspect | Details |
|---|---|
| Best entry channel for foreign brands | CBEC (fastest, lowest cost) |
| CBEC GACC registration required | ❌ No |
| General Trade GACC registration required | ✅ Yes |
| CBEC comprehensive tax (most supplements) | ~9.1% |
| General Trade tax (typical) | 20–35% |
| Blue Hat required for CBEC | ❌ No |
| Blue Hat required for General Trade | ✅ Yes |
| Blue Hat timeline | 2–5 years (registration) |
| Blue Hat cost | $50,000–200,000+ per product |
| CBEC Positive List | 1,476 HS codes (verify yours) |
| Key platforms | Tmall Global, JD Worldwide, Douyin Global |
| Order limit (CBEC) | ≤5,000 RMB/order; ≤26,000 RMB/year |
| Prohibited supplement ingredients | Veratrum, GBL, yohimbine, sibutramine derivatives |

China CBEC Reality Check
Is Your Supplement China-Ready?
Take the Supplement Trust Scorecard. Uncover weak spots in labels, claims, manufacturing proof, and social validation.
Disclaimer: This article is for informational purposes only and does not constitute legal advice. Regulations may change, and compliance requirements vary based on specific products and circumstances. Always consult with qualified legal and compliance professionals for guidance on your specific situation.
Last updated: April 2026






